Clinical Landscape
Lung Cancer
Lymphoma
Liver Cancer
Colorectal Cancer
Gastric Cancer
Nasopharyngeal Cancer
Biosyngen
Proprietary Technology Platform
Proprietary Technology Platform
-
M-CELTMAutomated and intelligent cell manufacturing platformImproved production cycle time of CAR-T/TCR-T product to 10-days. Manufactured in GMP facility in compliance to FDA and CDE requirements.
-
SUPER-T TMT cell safety optimization platformUnique T cell signaling tweaking strategy and TME-specific promoter to broaden tumor cell coverage while ensuring safety.
-
MSE-T TMT cell functional enhancement platformLibrary consisting of hundreds of motifs for T cell functional enhancement.
-
IDENTIFIERTMAntigen/Antibody/TCR discovery platformLarge tumor-specific TCR library for multiple high-frequency HLA haplotypes. Fast screening / optimization.
-
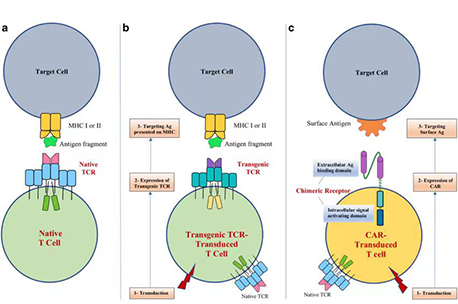
 T cells equipped with tumor detectorsWhen infused into the patient, the highly specific receptors enable detection and binding to the cancer cell, while importantly reducing the “on-target, off-tumor” toxicities that typically plague other immunotherapies. Taken together, this significantly boosts the clinical efficacy, reliability, and safety of BioSyngen therapeutics.
T cells equipped with tumor detectorsWhen infused into the patient, the highly specific receptors enable detection and binding to the cancer cell, while importantly reducing the “on-target, off-tumor” toxicities that typically plague other immunotherapies. Taken together, this significantly boosts the clinical efficacy, reliability, and safety of BioSyngen therapeutics.
-
 HIGHLIGHTS
HIGHLIGHTS First-in-class assetsBioSyngen’s proprietary cell therapy assets are First-In-Class Products against various solid cancers, filling the critical niches in immuno-oncology.
First-in-class assetsBioSyngen’s proprietary cell therapy assets are First-In-Class Products against various solid cancers, filling the critical niches in immuno-oncology. Safe by designRooted in a patient-centric approach, BioSyngen generates highly potent immunotherapies that are fit for each individual and carry enhanced safety profiles.
Safe by designRooted in a patient-centric approach, BioSyngen generates highly potent immunotherapies that are fit for each individual and carry enhanced safety profiles. -
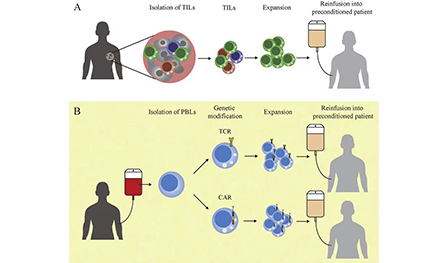
What is
CAR-T/TCR-T therapy?
LEARN MORE
Learn more about how we're advancing multiple therapeutic